
Seems very stupid QN doesn’t it.
Likewise,
Why water is used to extinguish fire. Why not use water in some conditions?
I want to give a very simple answer to the questions.

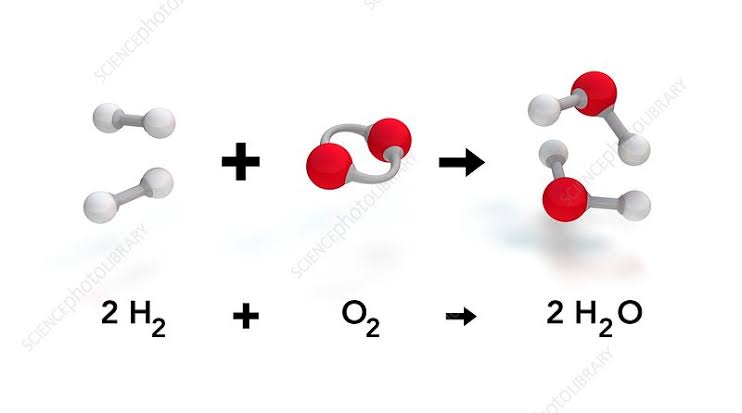
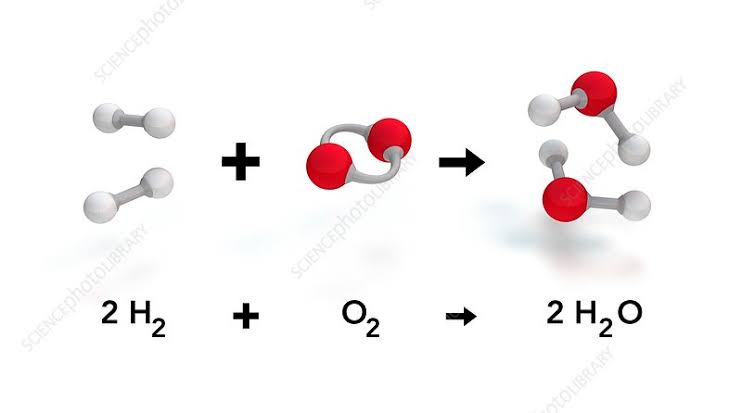
Water is a compound of hydrogen and oxygen. Hydrogen is a flammable element.
So is Oxygen.
But oxygen is said to be an oxidizing agent.
So why doesn’t water with these two elements burn?
That’s when we need to understand.
Water is produced by reaction between hydrogen and oxygen or by burning.
That means water is already burnt.
Now if you ask if it can be burned again, but another extreme oxidizing agent is required.
Here is another thing
Hydrogen and oxygen react to form water in an exothermic reaction.
There heat energy release is done.
Why water is used to extinguish fire.?
3 things are needed for fire to happen.
1.heat
2.fuel
3.oxygen
If any one of these is absent, fire does not exist.
When water is poured on fire, it absorbs its heat and extinguishes the fire.

This process is endothermic.
(heat absorbed from surroundings). Water will no longer be used in all types of fires.
One of them is electrical fire.
Because water is a conductor.
This will lead to more danger.
Here non-conducted substances have to be used.




Recent Comments