
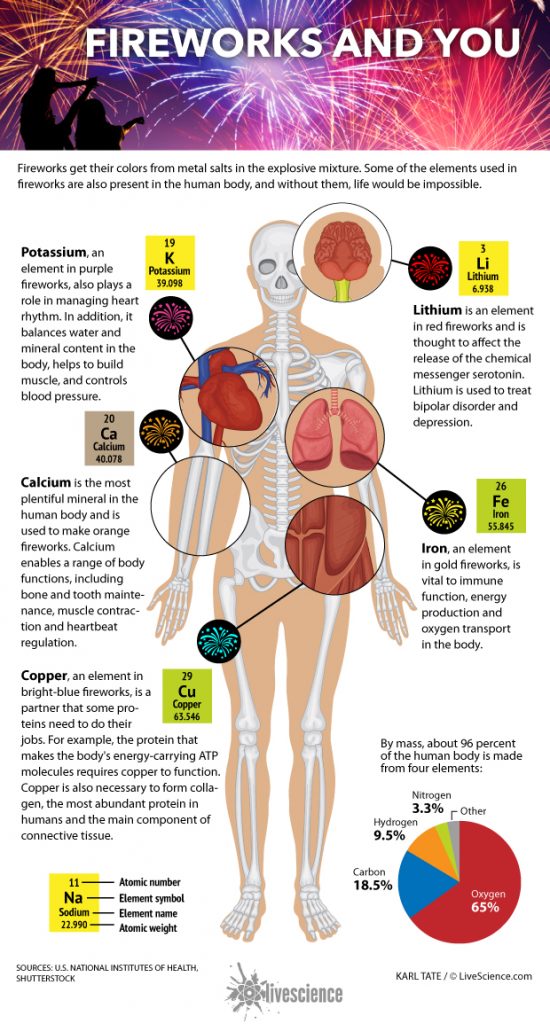
Our body is made up of four main elements. The four elements are oxygen, carbon, hydrogen and nitrogen. So, let’s see exactly what these components do.
That is, 65% to 67% of the human body’s oxygen is in the first place, because most of our body is water – about 50% to 60% – so most of the oxygen is “O” in H2O. Oxygen is crucial for energy production and metabolism or for the chemical reactions that take place within the body.

Next is carbon, which makes up 18% to 19% of the body’s weight, and is the major component of most life on Earth. It is an important building block of fats, carbohydrates and proteins, and the body absorbs most of the carbon from the food we eat.
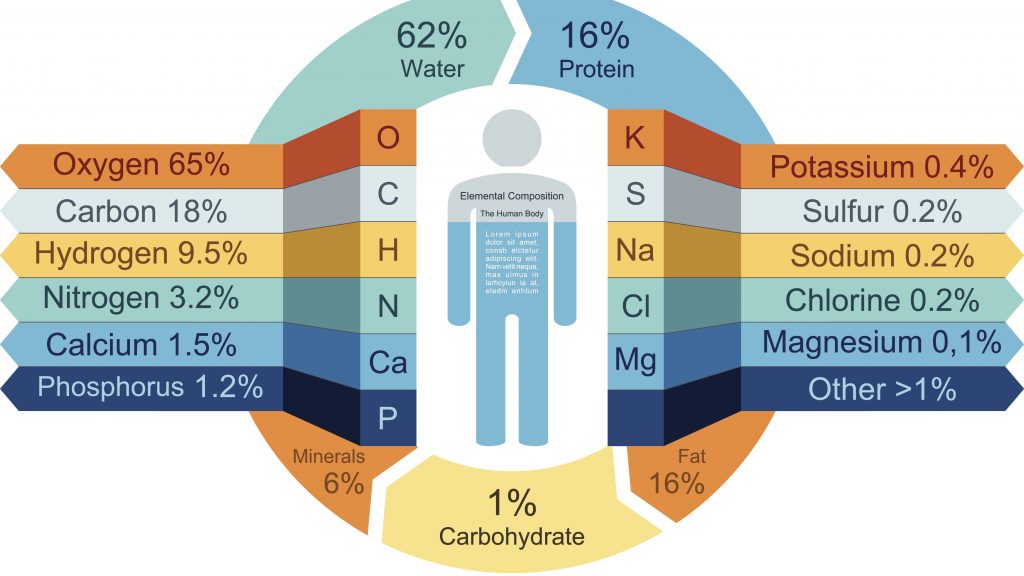
In third place is hydrogen. The human body contains more hydrogen atoms than any other element, but it makes up only 9% to 10% of our body mass. Hydrogen is another element in H2O, and each oxygen atom has two hydrogen atoms. In addition, hydrogen is an important component of proteins, carbohydrates and fats. Although hydrogen contains the largest number of atoms, its mass is much larger: oxygen and carbon have about 16 and 12 times the atomic weight of hydrogen, respectively. In other words, 16 hydrogen atoms are needed to match the mass of an oxygen atom, which is why hydrogen comes to one tenth of our mass.
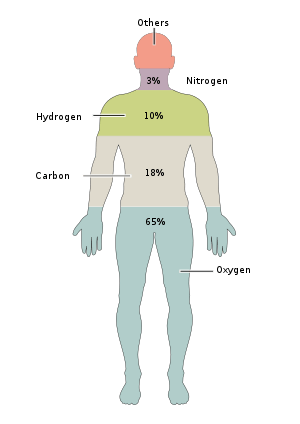
Nitrogen, the fourth most abundant gas in the Earth’s atmosphere, is estimated to be about 3%: Nitrogen is crucial for proteins and their building blocks, called amino acids. Nitrogen is an important component of DNA and RNA, including their nitrogen bases. Both contain cytosine, adenine, and guanine; In other words, without nitrogen, the cells in your body would not be able to store or copy genetic information.
In addition, calcium is the fifth most common bonus element in the human body, ranging from 1% to 2% by mass. According to the National Institutes of Health, 99% of that calcium is found in bones and teeth. That is, the complex body systems that keep us alive also have an incredibly simple elemental structure.




Recent Comments